iPLEX® Pro Sample ID Panel
Cost-Effective Genetic Fingerprinting
Toxicology screenings can be compromised by sample swapping, manipulation, and adulteration. Additionally, positive results are often questioned, leaving labs to justify their results.
Specimen validity testing (SVT) is a critical piece of toxicology screening and is regularly employed to determine if a urine specimen is abnormal or has been tampered with. However, standard specimen validity testing such as creatinine, oxidants, pH, nitrates, and specific gravity cannot detect certain methods of sample manipulation, such as urine substitution or synthetic urine, leading to inaccurate results.

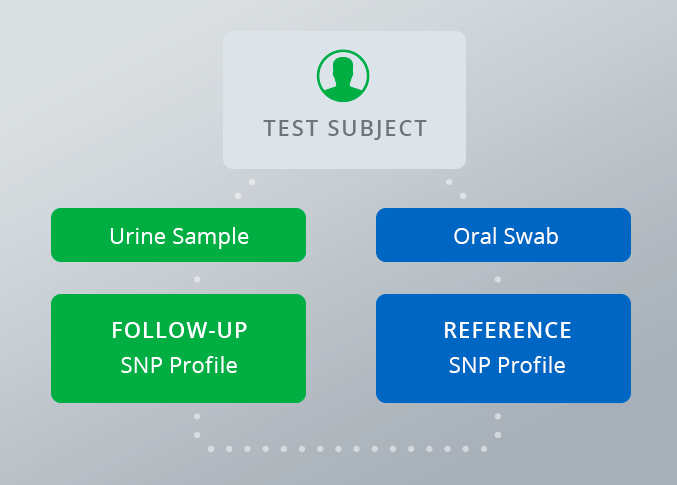
The iPLEX® Pro Sample ID panel provides more robust protection vs. traditional specimen validity testing. Using genetics, it accurately confirms positive results and detects fraudulent samples. The panel works with the MassARRAY® System to cost-effectively generate a unique genetic fingerprint for each sample. This fingerprint is used to match a test sample to the individual who submitted it, either confirming that it is valid or alerting the laboratory that a manipulation or mix-up has occurred.
Complete the form to receive our exclusive white paper: "Overcoming Standard Specimen Validity Testing Limits." Our team will also follow up to learn your goals!
To create each patient’s unique genetic fingerprint, a one-time oral swab is collected from the patient. Using 44 single nucleotide polymorphisms (SNPs), unique DNA patterns revealed by a PCR reaction, the panel maps a genetic profile for each subject. This profile is used to confirm their identity when providing subsequent urine samples. The panel is highly robust, with no less than a 1 in 45 billion chance of a false match.
Standard specimen validity tests account for appearance, temperature, pH, oxidants, creatinine, and specific gravity in urine samples. However, they fail to detect mixed, swapped, and synthetic urine samples and cannot confirm the identity when results are positive. With this panel, laboratories can supplement existing validity tests and may avoid observed sample collection, leading to certainty in results with a better experience, for all involved.
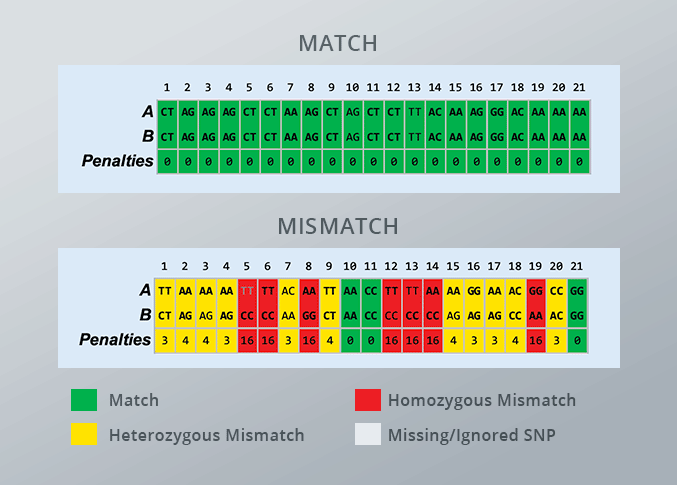
Once a test subject is entered into the Sample ID reference-matching software, laboratories receive clear results authenticating each urine specimen:
The MassARRAY technology is a benchtop platform that provides timely and accurate genetic analysis of up to thousands of samples in a single day. In addition to sample identification and validity testing, the platform can perform other applications such as pharmacogenetics, inherited disease, and oncology testing on the system. Ask for information regarding our other applications and custom panels.
The MassARRAY System workflow couples MALDI-TOF mass spectrometry with end-point PCR for highly multiplexed reactions under universal cycling conditions. Laboratories can consolidate molecular testing workflows on a single platform, with a simplified workflow and competitive cost per sample. Low reagent cost and automated reporting help save on disposable materials and labor costs.
Be confident in the authenticity of your results
Get started by completing the form at the top of this page, and we will respond within 24 hours to discuss your needs.
iPLEX Pro Sample ID Panel Brochure
View PDFLearn about the MassARRAY applications
Learn MoreWatch an overview of the MassARRAY System
Watch VideoAt Agena Bioscience, we are dedicated to advancing the impact of genomics in healthcare and precision medicine. Our highly sensitive and cost-effective platform, the MassARRAY® System, provides the capability for targeted genetic assays that satisfy the rigorous demands of genomic testing laboratories.